Introduction
In the fast-paced world of pharmaceutical manufacturing, efficiency, precision, and compliance are non-negotiable. HIGHNOW's High-speed Automatic Production Line for syringe/Ampoule/Vial/Oral Liquid/Cartridge represents the pinnacle of pharmaceutical packaging technology. This integrated solution combines blister packing and cartoning processes into a single, seamless operation, designed to meet the rigorous demands of modern pharmaceutical production.
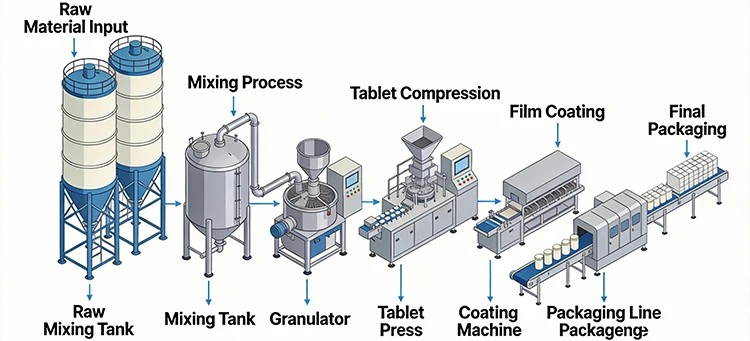
The production line is controlled by a sophisticated PLC system that integrates light, machine, electricity, and air technologies into an organic whole. This advanced automation enables continuous, fully automatic packaging of medicines, from Alu/PVC blister packing to automatic folding of medicine leaflets and cartoning.
Key Features and Benefits
Advanced Automation and Integration
- PLC Control System: The entire production line is managed by a programmable logic controller that ensures precise synchronization of all operations.
- Integrated Processes: Combines blister packing, leaflet folding, and cartoning into a single continuous process.
- Fully Automatic Operation: Runs continuously with minimal human intervention, reducing labor costs and increasing production efficiency.
High Production Efficiency
- Punching Frequency: 16-35 times per minute, with 1-12 plates per cycle
- Boxing Capacity: 200-350 boxes per minute
- Labor Savings: The entire line only requires 1-2 operators to manage the production process.
Advanced Quality Control
- Self-examining Function: Identifies broken blister covers, missing medicines, and other packaging defects.
- Empty Box Detection: Automatically detects and rejects empty boxes.
- 100% Waste Elimination: Ensures that only properly packaged products proceed to the next stage of production.
Premium Components and Design
- Imported Electrical Components: Over 90% of electrical components are sourced from famous international brands.
- Compact Structure: Integrates tray making, bottle filling, and cartoning into a single machine.
- User-friendly Operation: Designed for ease of use with intuitive controls and minimal training requirements.
Applications
The HIGHNOW High-speed Automatic Production Line is versatile and can handle various types of pharmaceutical products:
Prefilled Syringes
Ideal for packaging prefilled syringes with precise dosing requirements. The system ensures accurate handling and packaging of these delicate medical devices.
Cartridges
Designed to handle cartridge packaging for insulin pens and other injectable medications. The production line maintains the integrity of the sterile products throughout the packaging process.
Ampoules
Specialized handling systems ensure gentle processing of glass ampoules, minimizing breakage and contamination risks.
Vials
Accommodates various vial sizes and types, including those for injectable medications and lyophilized products.
Oral Liquid Bottles
Efficiently packages oral liquid medications in bottles, ensuring proper sealing and labeling.

Customization Options
HIGHNOW understands that every pharmaceutical manufacturer has unique requirements. That's why we offer customizable solutions to meet specific packaging needs:
Custom Blister Designs
- Tailored blister configurations for different product shapes and sizes
- Compatible with PVC, PP, APET, aluminum foil and other materials
- Flexible layout options to optimize packaging efficiency
Specialized Handling Systems
- Customized feeding mechanisms for unique product types
- Adjustable speed controls to match production requirements
- Integration with existing production lines
Compliance Solutions
- GMP-compliant design and documentation
- Validation support for regulatory requirements
- Cleanroom-compatible configurations
Quality Assurance and Compliance
GMP Compliance
The production line is designed and manufactured to meet the strictest GMP (Good Manufacturing Practices) standards. This ensures that pharmaceutical products are packaged in a controlled environment that maintains product integrity and safety.
Validation Support
HIGHNOW provides comprehensive validation support, including:
- Installation Qualification (IQ)
- Operational Qualification (OQ)
- Performance Qualification (PQ)
- Documentation for regulatory submissions
Quality Control Systems
- In-line inspection systems for defect detection
- Real-time monitoring of production parameters
- Statistical process control for consistent quality
After-sales Service and Support
HIGHNOW is committed to providing comprehensive after-sales support to ensure the smooth operation of your production line:
24/7 Online Support
Our technical support team is available around the clock to assist with any operational issues or questions.
On-site Commissioning
We provide on-site commissioning services to ensure proper installation and startup of the production line.
GMP Guidance
Our experts offer guidance on GMP compliance and help you maintain regulatory standards.
Spare Parts Availability
We maintain a comprehensive inventory of spare parts to minimize downtime in case of equipment failure.
Training Programs
We offer training programs for your operators to ensure they have the knowledge and skills to operate the production line effectively.
Frequently Asked Questions
How do you calculate and guarantee the Overall Equipment Effectiveness (OEE) for a high-speed syringe packaging line?
We engineer our lines for a demonstrable OEE of ≥87%, calculated through:
- Availability (A≥95%) via sub-20-minute format changeovers and predictive maintenance alerts
- Performance (P≥98%) maintaining speeds up to 400 units/min with <0.5% speed loss
- Quality (Q≥99.5%) using in-line vision systems with >99.98% defect detection
What specific validation methodology do you employ for 100% leak detection on ampoule blister packs?
We implement dual-validation methodology for critical leak testing:
- Primary detection uses high-resolution vacuum decay technology per ASTM F2338-09, capable of detecting leaks ≥5 microns
- Secondary verification is performed via weight-based mass comparison with precision ±0.01g
What is the maximum particulate contamination control level achievable for vial packaging operations?
Our integrated cleanroom-compatible design maintains ISO 14644-1 Class 5 standards at critical zones. Validation data shows <3 particles >0.5μm per m³ at the filling station, exceeding USP requirements for injectable products.
How do you prevent glass fracture during high-speed ampoule loading, and what's the acceptable breakage rate?
Our proprietary gentle-handling system maintains breakage rates <0.01% through:
- SUS304 stainless steel guides with Teflon coating
- Servo-controlled acceleration profiles limiting G-forces to <2G
- Vision-guided precision placement eliminating mechanical impact
What's the actual energy consumption profile per 100,000 units packaged, and how is power regeneration achieved?
Our regenerative drive system reduces energy consumption by up to 40% compared to conventional systems. The actual energy cost per 100,000 units is approximately €5.80 (based on €0.15/kWh), verified through ISO 50001 energy monitoring.
Conclusion
HIGHNOW's High-speed Automatic Production Line for syringe/Ampoule/Vial/Oral Liquid/Cartridge represents a significant advancement in pharmaceutical packaging technology. With its advanced automation, high production efficiency, and comprehensive quality control systems, this production line is designed to meet the evolving needs of the pharmaceutical industry.
Whether you're a large pharmaceutical manufacturer or a specialized contract packager, our production line offers the flexibility, reliability, and compliance you need to succeed in today's competitive market.
Contact HIGHNOW Today
To learn more about our high-speed automatic production line for syringe, ampoule, vial, oral liquid and cartridge packaging, contact our sales team today. We'll be happy to discuss your specific requirements and provide a customized solution that meets your needs.